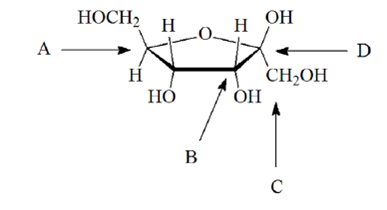
Left directed hydroxyl group (the mirror image) then represented the Pointed to the right, it was defined as a member of the D-family. If the hydroxyl group in the projection formula (farthest from the aldehyde group) was chosen by Fischer as the D / Lĭesignator site. The last chiral center in an aldose chain Six-carbon atoms) are shown below, with the asymmetric carbon atoms Projection formulas and names for the D-aldose family (three to To illustrate using present day knowledge, Fischer The mirror images of these configurations were then designated the L-family (+)-glucose and established a network of related aldose configurations Consequently, Fischer made an arbitrary choice for Click on this link for a review.Īt the time Fischer undertook the glucose project it was not possible to establish the absolute configuration Of chiral centers, that we now call the Fischer projection formula. To this end, he invented a simple technique for drawing chains Representing the configuration of each chiral center in an unambiguous One of the first tasks faced by Fischer was to devise a method of Received the 1902 Nobel Prize for chemistry for this accomplishment. His successful negotiation of the stereochemical maze presented by theĪldohexoses was a logical tour de force, and it is fitting that he Met in 1891 by the German chemist Emil Fischer. Pairs of enantiomers, and the initial challenge was to determine which The four chiral centers in glucose indicate there may be as many as sixteen (2 4) stereoisomers having this constitution. Stoichiometry of aldohexose cleavage is shown in the following equation. 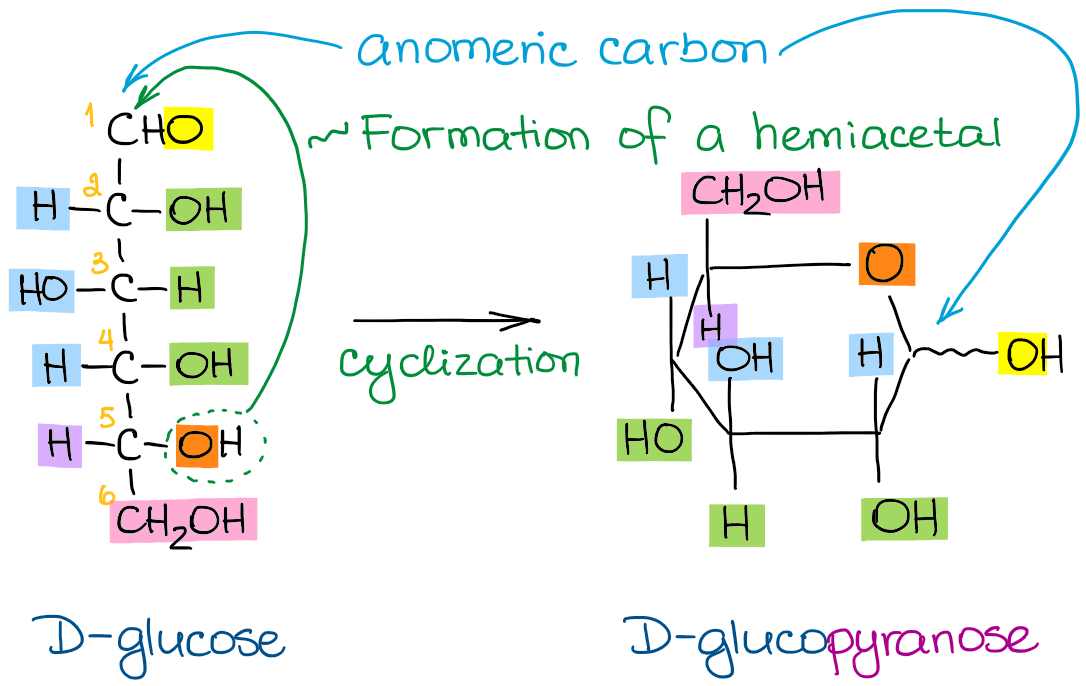
Is particularly useful for the analysis of selective O-substitutedĭerivatives of saccharides, since ether functions do not react. This oxidative cleavage, known as the Malaprade reaction Glucose and other saccharides are extensively cleaved by periodic acid, thanks to the abundance of vicinal diol moieties in their structure. The four middle carbon atoms in the glucose chainĪre centers of chirality and are colored red. It will change to display the suggested products and the gross Groups were assigned, one each, to the last five carbon atoms, because geminal hydroxyl groups are normally unstable relative to the carbonyl compound formed by loss of water. Groups, since a penta-acetate derivative could be made. The five oxygens remaining in glucoseĪfter the aldehyde was accounted for were thought to be in hydroxyl Somewhat stronger oxidationīy dilute nitric acid gave the diacid, glucaric acid, supporting the The mono-carboxylic acid, glucuronic acid. The hexa-alcohol sorbitol, also called glucitol, and mild oxidation to The presence of an aldehydeĬarbonyl group was deduced from cyanohydrin formation, its reduction to The six carbons are in an unbranched chain. Hot hydriodic acid (HI) was often used to reductively remove oxygenįunctional groups from a molecule, and in the case of glucose this The following diagram illustrates the kind of evidenceĬonsidered, although some of the reagents shown here are different from Glucose and many other aldohexoses was established by simple chemical The most common carbohydrate is glucose (C 6H 12O 6).Īpplying the terms defined above, glucose is a monosaccharide, anĪldohexose (note that the function and size classifications areĬombined in one word) and a reducing sugar. Sugars not oxidized by Tollens' or other reagents.Ĭarbohydrates have been given non-systematic names, although the suffix ose is generally used. Sugars oxidized by Tollens' reagent (or Benedict's or Fehling's reagents). Sugars having a ketone function or an acetal equivalent. Sugars having an aldehyde function or an acetal equivalent. Several classifications ofĬarbohydrates have proven useful, and are outlined in the following Or, if they are relatively small, sugars. Role, carbohydrates also serve as a structural material (cellulose), aĬomponent of the energy transport compound ATP, recognition sites on cell surfaces, and one of three essential components of DNA and RNA. The carbohydrates are a major source of metabolicĮnergy, both for plants and for animals that depend on plants for food.Īside from the sugars and starches that meet this vital nutritional N CO 2 + n H 2O + energy C nH 2nO n + n O 2Īs noted here, the formulas of many carbohydrates can be written as carbon hydrates, C n(H 2O) n, They originate as products of photosynthesis, an endothermic reductive condensation of carbon dioxide requiring light energy and the pigment chlorophyll. Carbohydrates are the most abundant class of organic compounds found in living organisms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
